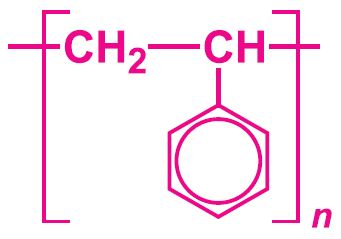
Uses of poly(phenylethene) (polystyrene)
The largest use for poly(phenylethene) is for packaging, particularly for foods such as poultry and eggs, for cold drinks and take-away meals.
Figure 1 Uses of poly(phenylethene).
It is also used in making appliances, including refrigerators, microwaves and blenders. It is the leading choice for jewel boxes (cases for CDs and DVDs) and is also widely used for its insulating properties.
Annual production of phenylethene (styrene)
The data given below are for the monomer, phenylethene (styrene). It is not only used to make poly(phenylethene) (polystyrene) itself but also for these important co-polymers:- phenylethene (styrene) with buta-1,3-diene, known as SBS
- phenylethene (styrene) with propenonitrile (acrylonitrile) and buta-1,3-diene, known as ABS (acrylonitrile-butadiene-styrene)
- phenylethene (styrene) with propenonitrile (acrylonitrile), known as SAN (styrene-acrylonitrile)
| World | 26.4 million tonnes1 |
| China | 13.2 million tonnes1 |
| US | 2.8 million tonnes2 |
Data from:
1. Estimate for 2016 Merchant and Research Consulting, 2013
2. Polystyrene 2015 Guide to the Business of Chemistry, American Chemistry Council, 2016
Manufacture of poly(phenylethene) (polystyrene)
Poly(phenylethene) is manufactured from its monomer, phenylethene. Phenylethene, in turn, is produced from benzene and ethene via ethylbenzene. There are thus three stages:
a) the manufacture of ethylbenzene from benzene
b) the manufacture of phenylethene (styrene)
c) the polymerization of phenylethene (styrene)
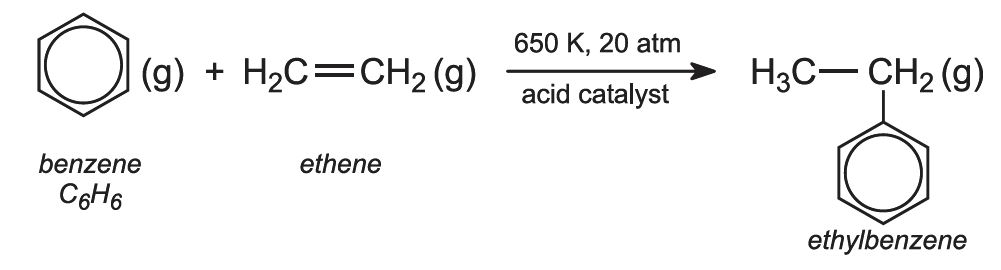
(a) The manufacture of ethylbenzene from benzene
Benzene vapour and ethene are mixed and passed over an acid catalyst, at 650 K and 20 atm pressure:
This is an example of a Friedel-Crafts reaction. The acid catalyst now used is a zeolite, ZSM-5, an aluminosilicate.
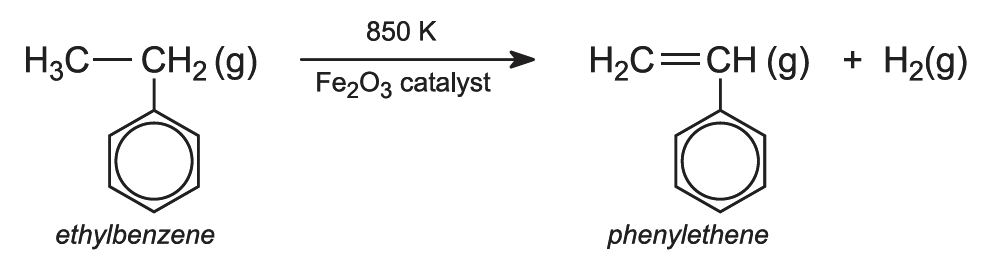
(b) The manufacture of phenylethene (styrene)
Ethylbenzene vapour is mixed with excess steam and passed over heated iron(lll) oxide. Other metal oxides used as the catalyst including those of magnesium, chromium(III) and zinc, usually coated on carbon or alumina. It is dehydrogenated:
A small amount of potassium oxide is mixed with the iron(lll) oxide (which keeps the catalyst in the iron(lll) state).
The steam reduces 'coking' (the formation of soot on the catalyst from the decomposition of ethylbenzene at the high temperatures used).
(c) The polymerization of phenylethene (styrene)
Radical polymerization is used to produce poly(phenylethene) (polystyrene). The process is an example of addition polymerization.
The predominant polymerization technique is continual thermal mass polymerization which is initiated by heat alone. Suspension polymerization is also used. This technique requires the use of an initiator such as dibenzoyl peroxide.
Poly(phenylethene) is a clear thermoplastic, with good moisture resistance, but is rather brittle. A tougher product is also manufactured by polymerizing phenylethene containing 5-10% dissolved poly(buta-1,3-diene) rubber. This tougher product - generally knownas High Impact Polystyrene (HIPS) - is made exclusively by continuous thermal mass polymerization, in which heat is required to initiate the polymerization reaction. This toughened polymer is translucent.
The structure of poly(phenylethene) made by these technologies is completely random in structure and is known as an atactic structure. By modification of the polymerization technique - principally by the use of metallocene catalysts - stereoregular (syndiotactic) structures can be obtained. This syndiotactic polymer (sPS) has improved properties - particularly thermal and mechanical.
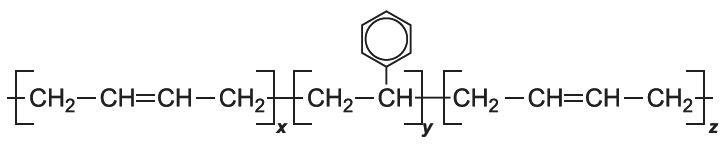
A very important co-polymer is produced from phenylethene (styrene) is with butadiene, SBS, much used in car tyres.
SBS is an example of a block co-polymer. First, phenylethene is polymerized. Buta-1,3-diene is then added and adds on to both the reactive ends of the poly(phenylethene) molecules to form SBS:
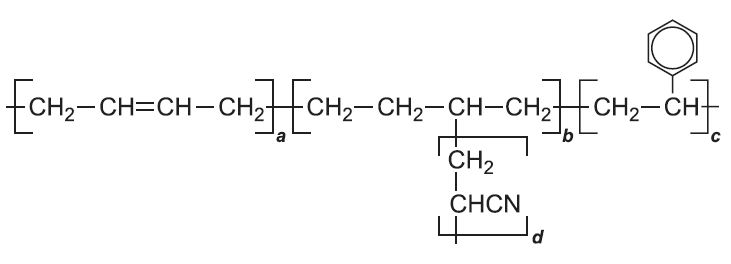
A further modification involves the co-polymerization of phenylethene (styrene) with propenonitrile (acrylonitrile) in the presence of poly(buta-1,3-diene) to make ABS plastics. A, B, S represent acrylonitrile, butadiene and styrene, which give strength (A), flexibility (B), and hardness (S). Typically this plastic has a composition: 60% (w/w) phenylethene (styrene), 25% propenonitrile (acrylonitrile), 15% buta-1,3-diene. The initiator used is often potassium peroxydisulfate, K2S2O8.
This is an example of a graft co-polymer. The backbone of the polymer is formed from phenylethene (styrene) and buta-1,3-diene. Propenonitrile (acrylonitrile) is added to the system and forms a grafted side chain onto the backbone. The nitrile adds to the double bond on the butadiene unit:
|
ABS is tougher, scratch proof and more chemically resistant than rubber-modified poly(phenylethene) and is used, for example, in casings for computers, cycle helmets, calculators, telephones, vacuum cleaners and toys.
Another co-polymer is formed on polymerizing a mixture of phenylethene (styrene) and propenonitrile (acrylonitrile). It is known as SAN (styrene-acrylonitrile). It is less flexible, more transparent and has more resistance to heat and chemicals than poly(phenylethene). It is used in car headlamps, cassette covers, syringes and high quality kitchen appliances.
Often SAN is blended with ABS to make it an even more rigid polymer.
Another variation is the co-polymer formed between ABS and methyl 2-methylpropenoate, which has a high resistance to chemical attack, high transparency and is very tough.
Poly(phenylethene) accounts for 50% of the polymers producede from phenylethene and SBS,ABS and SAN, a further 27%, 11% and 1% respectively1.
1. The Styrene Forum, 2012
Expanded poly(phenylethene) (polystyrene)
Expanded poly(phenylethene) is manufactured as beads containing pentane (a liquid at room temperature).
When they are heated in steam, the hydrocarbon volatilises and the beads expand (Figure 3). These are subsequently blown into moulds and fused by further steaming and then cooling. The expanded poly(phenylethene) has good thermal insulation and shock absorbing properties.
|
Date last amended: 24th April 2017