A composite is made by physically combining two or more materials (components) to produce a combination (blend) of structural properties not present in any individual component. They can, for example, provide greater strength and rigidity than is found in any of the separate components while being as light as possible.
Composites are being increasingly developed for a multitude of tasks. For example, fibre reinforced composites are being used to replace materials such as metals and their alloys.
Composites are designed to offer:
- low weight
- stiffness and strength
- low coefficient of expansion
- resistance against fatigue
- ease in manufacturing complex shapes
- simple repair of damaged structures
- resistance to corrosion
There are many different types of composites. This unit looks at two which are of major importance and which are currently being developed for many uses:
- Fibre reinforced polymer composites
- Particle reinforced composites
Phases in composites
Fibre and particle reinforced composites usually consist of a phase which is more or less continuous. This continuous phase is also known as the matrix, and the material that is distributed through the matrix is known as the dispersed phase (Figure 1).

Figure 1 Illustrating the phases of a composite.
The dispersed phase is sometimes called reinforcement if it is a phase added to increase strength. Alternatively, it is called a filler if it is added for other purposes, for example to bulk up the matrix at low cost without affecting the properties of the composite. There may also be a phase to create a bond between layers or phases, sometimes called the interface.
When designing composites the following factors must be considered:
- The proportion of matrix and dispersed phase can vary depending on the intended use of the composite. The matrix phase can be the bulk material with particles or fibres dispersed through it. It can also be the minor phase, more like glue cementing the particles or fibres together, providing stiffness to otherwise very flexible fibre arrays and giving a structure to what would otherwise be loose particles. The matrix also serves to protect the dispersed phase from mechanical damage and chemical attack. Where the matrix is ductile, it prevents propagation of cracks between fibres or particles, even when it is subjected to hard wear.
- The size and shape of the particles and fibres of the dispersed phase govern how closely they can be packed. Although smaller particles provide more surface area for contact with the matrix, longer fibres give better reinforcement. There is a balance to be struck.
- The interface between matrix and dispersed phase also controls the overall performance of the composite. The strength of a composite depends not only on the properties of the matrix but on how well it sticks to the particles and fibres of the dispersed phase.
Fibre reinforced polymer composites
Fibre reinforced polymer composites (FRPs, also known as polymer matrix composites (PMCs)) are subdivided into:
a) Carbon fibre reinforced polymer composites (CFRPs)
b) Glass fibre reinforced polymer composites (GFRPs)
c) Aramid fibre reinforced polymer composites (AFRPs)
In each case the fibre is encased in a resin matrix (the continuous phase). These matrices are usually acrylicepoxyphenolic or polyester resins.
Manufacture
Manufacturing a fibre-reinforced composite involves several steps in which the filler (fibre) is produced and then, if required, aligned prior to the introduction of the matrix.
Fibres produced from polymers (for example, the aramids, see below), glass and metals can be produced from the molten state by drawing them. Most carbon fibres are first drawn from a polymer, for example poly(propenonitrile) (polyacrylonitrile), which is then oxidized, followed by pyrolysis. If the fibres are unaligned (random orientation) with each other, the filler and matrix can be blended together as powders or slurries, shaped or moulded, and the matrix hardened and bonded to the filler by heat or by chemical reaction. The shaping may be accomplished by injection moulding or casting of the composite. If required, additional machining is done before any further treatment to harden the composite.
The production of aligned fibre (Figure 1) composites is more complicated. The fibres may be used as monofilaments, or they may be twisted into yarns of up to 10 000 filaments. The yarns are then woven or knitted into two or three-dimensional fabrics and can be then formed into tapes by weaving or braiding. Filaments can also be processed into non-woven mats of fibres, randomly orientated in two dimensions.
The mats, tapes and fabrics may be impregnated with matrix material (or materials from which the matrix is made) before final assembly and processing, and are then referred to as a prepreg (pre-impregnated).
Thus, there is no need to handle the individual chemicals. It is simply rolled out and used as a laminate. Curing is accomplished by heating.
The fabrics and mats are placed in moulds, sometimes by hand, and impregnated with matrix material and processed. They can be compressed mechanically or by pressure or vacuum forming techniques. Filaments or tapes can also be wound, passing through a bath of matrix before being processed into a solid shape.
A simple example of a prepreg is the bandage that is used to set broken limbs (Unit 67). The bandage is made of a polyester and it is impregnated with a linear polyurethane, the matrix. The bandage, the prepreg, is flexible and is wound around the broken limb. On soaking, the polyurethane molecules react with water to form cross linkages between the molecules, producing a strong but light cast.
(a) Carbon fibre reinforced polymer composites (CFRPs)
Carbon fibres are generally made by heating filaments of poly(propenonitrile) (polyacrylonitrile, PAN) at ca 500 K, under an atmosphere of air to form oxidized PAN. The oxidized PAN is then placed into a furnace with an inert atmosphere of a gas such as argon, and heated to ca 2000 K, a process known as pyrolysis, the heating of a substance in the absence of air. The product, carbon fibre, contains chains of carbon atoms which are bonded side-to-side (ladder polymers), forming narrow sheets of carbon atoms, one atom thick, known as graphene. The isolation of graphene, by Professors Andre Geim and Konstantin Novoselov at the University of Manchester, is so important that the two scientists were awarded the Nobel Prize in Physics in 2010.
One can imagine it as a piece of chicken-wire on a very small (atomic) scale. Graphene is also the basic structure of graphite and of carbon nanotubes. In the case of a carbon fibre, the sheets merge to form a single circular filament. Carbon fibres are used as a filler in a continuous matrix of a polymer, often an epoxy resin. Layers of carbon fibre cloth are laid down in the shape required, usually in a mould which is then filled with epoxy resin and heated.
Uses
Carbon fibre composites are relatively expensive construction materials and therefore used when their properties of lightness and strength are of paramount importance. Examples of use include high quality sports equipment, such as tennis racquet frames, golf clubs and fishing rods, and in laptops and cameras.
They are also used extensively in the construction of aircraft. The fuselages of the newest commercial aeroplanes (the Boeing 787 (Dreamliner) and the Airbus A350) are constructed mostly from CFRP for its superior lightness and strength.
Another property of CFRPs is exploited in aircraft brakes. These are required to absorb considerable quantities of energy rapidly without mechanical failure or seizure. The usual construction is based on multiple rotating and stationary discs, which can reach surface temperatures of up to 3000 K. The disc material must therefore have excellent thermal and shock resistances and high-temperature strength, together with good thermal conductivity. Carbon is an ideal material and the discs are made from a carbon composite where the filler is carbon fibre and the matrix is carbon produced from the pyrolysis of methane. Additionally, weighing some 30% less than steel discs, they save a considerable amount of fuel.
CFRPs have also been used for many years to construct the body of F1 racing cars, giving drivers greater protection even in crashes at over 300 km h-1. They are now being used in luxury cars (part of the Mercedes Benz range, and for the roof of the GM Corvette ZR1) and as protective gear for motor cyclists.
CFRPs are increasingly used to 'retrofit' existing large structures such as bridges made of reinforced concrete. The carbon fibre fabric is wrapped around the parts that need strengthening.
(b) Glass fibre reinforced polymer composites (GFRPs)
Glass fibre is made from silica (sand), sodium carbonate and calcium carbonate together with other compounds to give the specific properties required. The materials are heated to ca 1700 K in a furnace and then extruded directly from the furnace through metal (a platinum/rhodium alloy) orifices of various diameters (4 - 34 urn) to produce filaments. A high speed winder revolving faster than the exiting molten glass draws them under tension into very fine filaments. The number of orifices varies from 200 - 8000.
The filaments are coated with a lubricant to protect them and bundled together on a drum, looking like a spool of a thread.
In fibreglass, in which glass fibres are the filler (dispersed phase), the matrix is usually a polyester resin although epoxy and acrylic polymers are also used.
Uses
GFRPs are used widely in the manufacture of boats for reasons of cost and maintenance. While large ships are usually constructed in steel, over 80% of marine hulls, less than about 40 m in length, are made of glass fibre reinforced polymer. This is a much cheaper process and the hull is easier to maintain. Additionally, there are certain applications in which the magnetic, electrical or thermal properties of GFRPs are advantageous, an example being minesweepers which need to be non-magnetic in order to avoid activation of mines.
Glass-phenolic resin prepregs are also used to improve protection for armoured vehicles.
(c) Aramid reinforced polymer composites (ARPCs)
An aramid is a polymeric aromatic amide. Kevlar® is a particularly widely used aramid.
It is very difficult to handle as the only effective solvent is concentrated sulfuric acid. Kevlar® is made from lightweight atoms but is very strong and flexible, weight for weight five times as strong as steel. Its strength comes from the way the polymer chains are aligned, the flat molecules being held together by hydrogen bonds. These sheets of molecules can stack up along the fibre axis (Figure 2).
These fibres, already strong, are used as the filler in ARPCs with a phenolic resin or epoxy resin as a matrix. They are particularly useful where energy has to be absorbed and dissipated, and they are also able to resist abrasion.
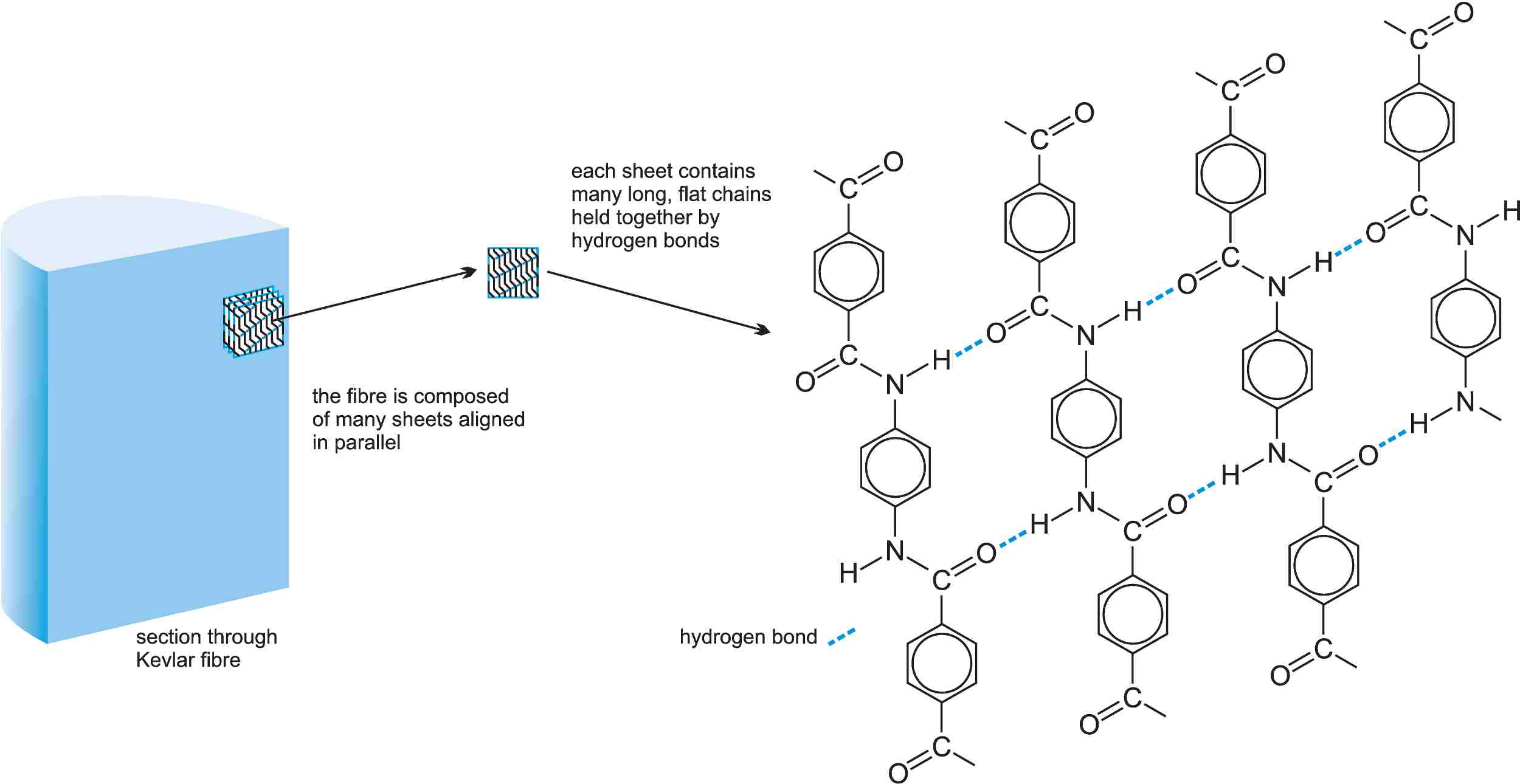
Figure 2 The structure of Kevlar®.
Uses
These composites have been used widely in aviation, for helicopter rotor blades, in sport, to make tennis, badminton and squash racquets, and in boats such as kayaks and dinghies.
|
Figure 3 Kevlar® and other polyaromatic amides are also used as fibres in fabrics for protective clothing. Their fire resistant properties lead to their use by firefighters and their strength means they are used for body armour.
|
 |
Particle Reinforced Composites (PRCs)
There are essentially two types of particle reinforced composites, large particle reinforced and small particle reinforced composites. It is not strictly the physical dimensions of the particles by which the materials are classified, rather it is the mechanism of reinforcement. In a small particle reinforced material the mechanism is on a molecular level and the particles may be dispersed into or precipitated from the matrix.
Large particle reinforcement, as the name suggests, involves larger particles and a distribution of the load between phases. Whatever their geometry the particles are small relative to the size of the structure and evenly distributed through it.
The particles may improve the fracture strength of the composite over that of the matrix by preventing or impeding crack propagation through the matrix, either physically blocking and stopping cracks or diverting and splitting them so as to hamper their progression across the piece. They may also improve the stiffness and strength of the composite over that of the matrix, by carrying a proportion of the load.
Uses
Concrete is a simple, everyday example of a PRC, the composition varying according to its intended use. The most common matrix is a Portland cement paste, which undergoes chemical reactions leading to setting in a few hours with hardening continuing over a number of weeks or months. This material is reinforced with aggregate (the dispersed phase) such as pebbles or chippings (1-2 cm) and sand (1-2 mm). In addition to reinforcement, the dispersed phase brings other benefits. The initial setting reactions of the concrete are exothermic and, when only cement and water are used, this leads to problems with heat dissipation and cracking. The addition of the aggregate ameliorates this problem by acting as a heat sink reducing the rate and magnitude of the temperature increase.
Concrete often contains an additional phase in the form of steel wire, rods or cable to give even greater strength.
Refractory carbides are hard, wear-resistant ceramic materials such as titanium and tungsten carbides (TiC and WC). They can be incorporated into a matrix of metal, often cobalt or nickel, to make machine tool cutting tips. This is an example of a cermet, a combination of ceramic and metal. The carbide provides the wear resistant cutting edge, but by itself would shatter on impact with the metal being shaped. The metal matrix adds resilience and is easier to manufacture. Over time, as particles are dislodged, fresh ones are exposed to retain the sharp cutting edge, making the tool to some extent self-sharpening.
Carbon black essentially consists of spherical particles of carbon produced by combustion of oil or gas with a limited supply of oxygen, and is often known as furnace black. The most important use of the particles is as a particulate reinforcing phase in rubber used in the manufacture of tyres. Tyres can be made from different blends of natural rubber and synthetic rubbers, the blend depending on its intended use (for example for the side wall, the tread or inner lining).
The synthetic rubbers include:
- poly(buta-1,3-diene)
- poly(2-methylbuta-1,3-diene) (polyisoprene)
- co-polymers of phenylethene (styrene) and but-1,3-diene
- the brominated co-polymers of 2-methylpropene (isobiutene) and isoprene
Using carbon black in the rubber has improved wear resistance. However, for the reinforcement to be effective, the particles must be in the range 20-50 nm, evenly distributed in amounts up to 30%, and well bonded to the matrix. Although more expensive to produce, the tyres made from this composite have greater longevity.
Date last amended: 18th March 2013
