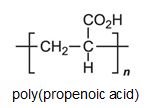
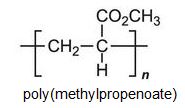
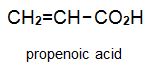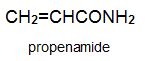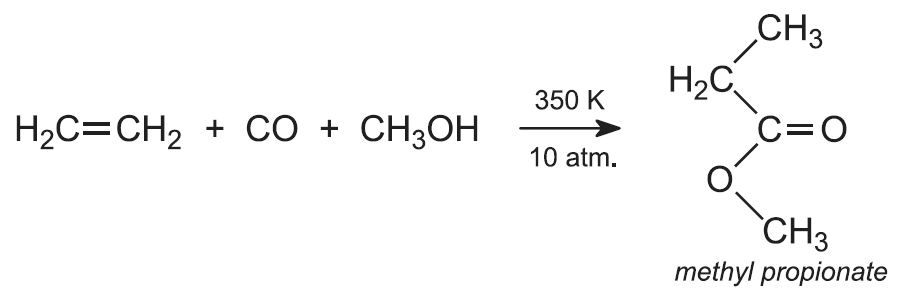There is a group of polymers, the acrylics, which can be regarded as based on acrylic acid, more formally named propenoic acid.
The acid polymerizes by addition polymerization to poly(propenoic acid) (polyacrylic acid).
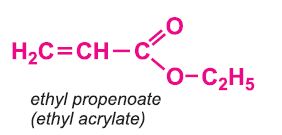
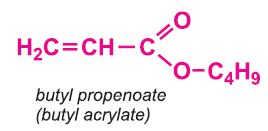
Other compounds, based on the acid, which form acrylics include the methyl, ethyl and butyl esters of propenoic acid.
 |
 |
 |
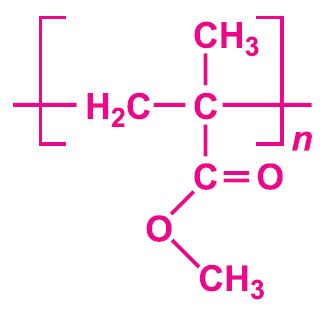
Each of these esters can be polymerized, for example the methyl ester forms this acrylic:
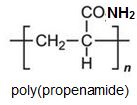
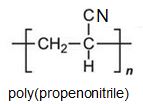
Two other important monomers, based on the acid, are propenonitrile (acrylonitrile) and propenamide (acrylamide).
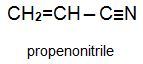 |
|
It can be seen that the polymers have the basic structure of the acrylics shown above.
 |
|
Poly(methyl 2-methylpropenoate, (poly(methyl methacrylate)), is another important polymer and is known simply as acrylic or acrylic glass. It is based on the methyl ester 2-methylpropeonic acid (methacrylic acid).
This website contains three units on the acrylics:
Poly(propenoic acid) (Polyacrylic acid)
Poly(propenonitrile) (Polyacrylonitrile)
Poly(methyl 2-methylpropenoate) (Poly(methyl methacrylate))
Uses of poly(methyl 2-methylpropenoate) (polymethyl methacrylate)
Poly(methyl 2-methylpropenoate) is better known as Lucite, Perspex and Altuglas (when in sheet form) and as Diakon (when in powder form).
The cast sheet is used in baths and other sanitary ware, which along with illuminated signs, is the largest use of the polymer. High molecular mass cast sheet (Perspex) is also used as a lightweight replacement for glass. Lower molecular mass products, made by suspension or solution polymerization (Diakon), are used in car lights and domestic lighting.
Special grades are used in diverse applications such as false teeth and eyes and as a major component of bone cements.
The monomer is used in adhesives, surface coatings and in paints.
Figure 1 Uses of poly(methyl-2-methylpropenoate).
Annual production of poly(methyl 2-methylpropenoate) (polymethyl methacrylate)
Polymer
| World | 2.9 million tonnes1, 2 |
1. Estimate for 2020, Grand View Research, 2015
2. It is estimated that over 0.5 million tonnes of the polymer will be bio-based. Global Market Insights, 2016
Monomer
| World | 4.8 million tonnes3 |
3. Estimate for 2020, Global Industry Analysts Inc. 2016
Manufacture of poly(methyl 2-methylpropenoate) (polymethyl methacrylate)
(a) The monomer
The monomer is the methyl ester of 2-methylpropenoic acid, methyl 2-methylpropenoate (methyl methacrylate):
Currently, most of the monomer is made by two processes
(i) From propanone (acetone)
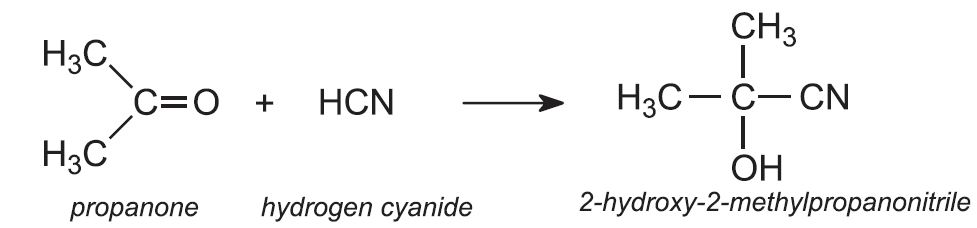
Worldwide, over 80% of the monomer is made from propanone by a sequence of steps which begins by reacting propanone with hydrogen cyanide.
| Figure 2 The hydrogen cyanide is manufactured from ammonia and methane. A mixture of the gaseous reactants is passed through a platinum-rhodium gauze catalyst. The heat generated by the reaction keeps the gauze glowing red at 1270 K. The photograph was taken through a sight glass located on the reactor. |  |
Propanone and hydrogen cyanide form 2-hydroxy-2-methylpropanonitrile:
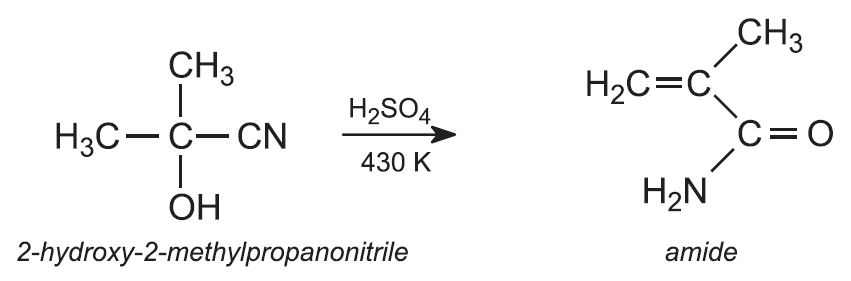
This product, on reaction with concentrated sulfuric acid at about 430 K, is dehydrated and the nitrile goup (CN) hydrolyzed to the amide. This is a step-wise process involving both dehydration and hydrolysis. The reactions can be summarised as:
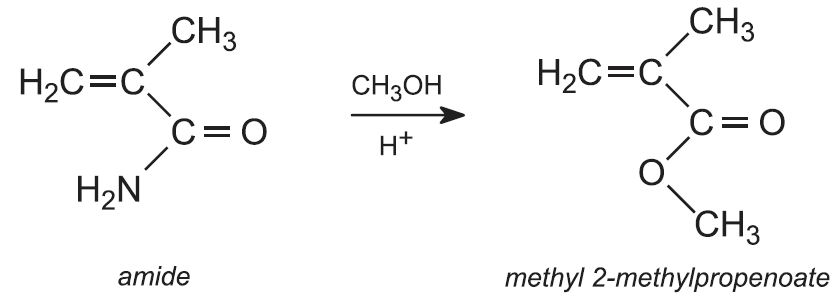
The temperature is decreased to 370 K and methanol is added. The amide group is hydrolyzed and esterified. The reactions can be summarised as:
The product is continuously removed by steam distillation.
A drawback to the process is the co-production of ammonium sulfate. Together with 'spent' sulfuric acid from the reactions above, the ammonium salt is heated strongly in oxygen in a furnace, in a process similar to that used for recycling sulfuric acid. The products formed are nitrogen, carbon dioxide and sulfur dioxide. The latter is then converted to sulfuric acid using the Contact Process. The use of pure oxygen reduces the size of the furnace which saves on both energy and equipment costs.
Research is being carried out to find efficient methods for the production of bio-based propanone from carbohydrates by the ABE process.
(ii) From ethene, carbon monoxide and methanol
Much work has been done to find alternative sources of the monomer and a promising route, which is now in use, uses a mixture of ethene, carbon monoxide and methanol in the liquid phase under pressure of about 10 atm at 350 K:
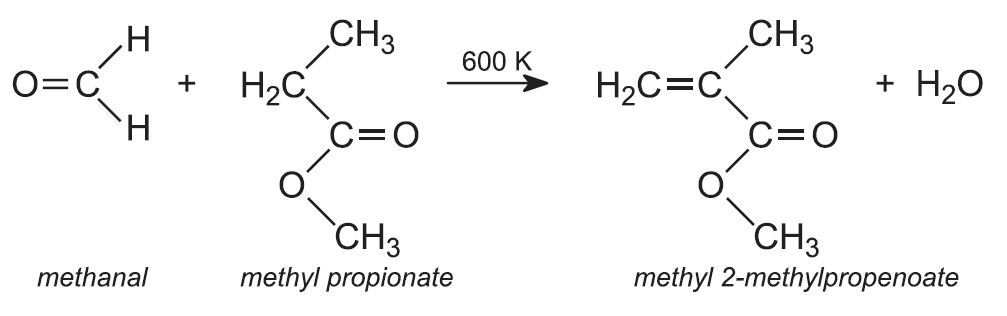
The resulting ester, methyl propionate, is reacted with methanal to form methyl 2-methylpropenoate. A fixed bed reactor is used and the reactor and catalyst (for example, caesium hydroxide on silica) are heated to 600 K:
 |
| Figure 3 The building of the first site for the manufacture of methyl 2-methylpropenoate, the monomer used to make poly(methyl 2-methylpropenoate), from ethene, carbon monoxide and methanol. The site is on Jurong Island just off the shore of Singapore. 1 The reactor to make methyl propanoate from ethene, carbon monoxide and methanol. 2 The reactor to make methyl 2-methylpropenoate frpm methyl propanoate and methanal. 3 Storage vessels. 4 Section being built for drying methanal, produced in aqueous solution from methanol. 5 Section being built to handle waste gases. 6 Drying column ready for installation at 4. 7 Control room for the site. 8 E;ectrical switch room. 9 Section being built for the purification of methyl 2-methylpropenoate. |
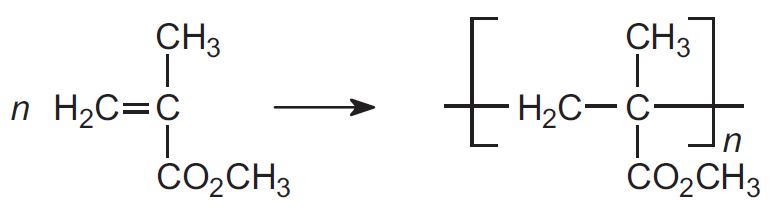
(b) The polymer
Polymerization of methyl 2-methylpropenoate is achieved by a free radical process using an initiator, such as an azo compound or a peroxide:
The amount of initiator employed affects both polymerization rate and resulting molecular mass of the polymer.
Polymerization is carried out commercially in several ways, i.e. in bulk, solution, suspension and emulsion.
The process is an example of addition polymerization.
(c) Co-polymers
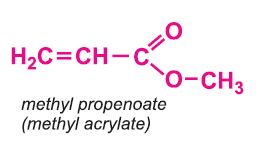
Co-monomers are often used together with the methyl 2-methylpropenoate. For example, most commercial grades of poly(methyl 2-methylpropenoate) used in injection moulding or extrusion applications contain a small amount (ca. 4%) of co-monomer, such as methyl propenoate (methyl acrylate) (when casting sheets of the polymer) and ethyl propenoate (ethyl acrylate) (when extruding sheets of the polymer).
In these co-polymers, the monomers are randomly arranged. The resulting polymers have increased thermal stability compared to the homopolymer.
With butyl propenoate (butyl acrylate), a co-polymer is produced which is used as a base for emulsion paints.
It is also co-polymerized with ABS to produce a very tough polymer which is both rigid and has excellent clarity. It is used, for example, in medical applications and in cosmetic packaging.
Date last amended: 11th January 2019