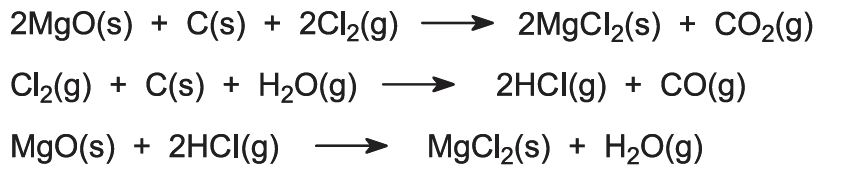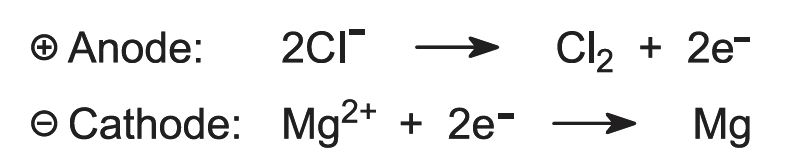Uses of magnesium
Magnesium is the third most used metal in construction (after iron and aluminium).
Nearly 70% of the world production of magnesium is used to make alloys, which have a very low density, comparatively high strength and excellent machinability. These alloys contain one or more of the elements aluminium, zinc, manganese or silicon in various amounts, depending on how the alloy is to be processed.
Half of these alloys are used to make die castings with about 90% magnesium. Car components such as steering wheel cores, gearbox casings, dashboard structures and radiator supports are often made from high pressure die cast magnesium alloys.
Zirconium and rare earth elements are added in some alloys to make the alloy stronger. This group of alloys is normally sand-cast into parts such as helicopter gearboxes and jet engine auxiliary gearboxes. Some high performance cars are made of a magnesium alloy as are casings for cameras.
Another half of the magnesium used in alloys is as an alloying additive, in the aluminium industry. The alloys are used in packaging, particularly in drinks (beverage) cans and in foil to protect food.
Most metal beverage cans manufactured in the United States are made of the aluminium alloyed with about 5% magnesium and a small amount of other elements. In Europe and Asia, the metal can contains about 50% steel and 50% aluminium alloy with the top being aluminium alloy.
Magnesium alloys are also used as sacrificial anodes. When connected to a less reactive metal, the magnesium becomes the anode of an electrical cell, and corrodes in preference to the other metal. This is used to protect the hulls of steel ships and the under-water structure of oil platforms and pipelines from corrosion.
Another very important use of magnesium is in the manufacture of titanium. About 10% of the world production of magnesium is used in this way.
Another 10% is used in the manufacture of high grade steel for constructions, such as large buildings and bridges. It is added in the molten state to molten iron, to remove sulfur by chemical reaction, the slag of magnesium sulfide being skimmed off.
Perhaps one of the best known but smallest uses of magnesium is in distress flares, fireworks and other incendiary devices. They contain very small pieces of magnesium which can be ignited.
Annual production of magnesium
These figures are for primary production from the ore and do not include secondary production from recycled materials.
| World | 910 000 tonnes1 |
| China | 800 000 tonnes1 |
| U.S. | 70 000 tonnes2 |
| Russia | 30 000 tonnes1 |
| Israel | 25 000 tonnes1 |
| Kazakhstan | 20 000 tonnes1 |
Data from:
1 U.S. Geological Survey, Mineral Commodity Summaries, 2016.
2 Last available figure is 2012 (Minor Metals Trade Association, 2012)
In 1993, the amount of magnesium produced in China was negligible (ca 5%), twenty years later, China now manufactures nearly 90% of the world's magnesium. Although the country has rich deposits of appropriate magnesium ores, it was the rapid economic growth that led to increased demand in the country for products using magnesium alloys. This in turn resulted in the shut-down of plants in many countries. The manufacturing processes use very large amounts of energy, and for this reason production in many countries is uneconomic. Magnesium is not now produced in Western Europe.
Manufacture of magnesium
Magnesium is found in solution in sea-water (about 1.3 kg m-3 magnesium) and in natural brines. It is also found extensively in the ores magnesite (MgCO3) and dolomite (MgCO3.CaCO3).
Magnesium is principally produced by two methods:
a) thermal reduction of magnesium oxide
b) electrolysis of magnesium chloride
Before the expansion of production in China, electrolysis was the more common method of production in countries where electrical energy is produced relatively cheaply. Most Chinese plants, however, use an updated version of the thermal reduction process originally developed in Canada in the 1940s to boost production during World War II (the 'Pidgeon Process').
(a) Thermal reduction process
Dolomite ore is crushed and heated in a kiln to produce a mixture of magnesium and calcium oxides, a process known as calcining:
The next step is reduction of the magnesium oxide. The reducing agent is ferrosilicon (an alloy of iron and silicon) which is made by heating sand with coke and scrap iron, and typically contains about 80% silicon.
The oxides are mixed with crushed ferrosilicon, and made into briquettes for loading into the reactor. Alumina may also be added to reduce the melting point of the slag. The reaction is carried out at 1500 - 1800 K under very low pressure, close to vacuum. Under these conditions the magnesium is produced as a vapour which is condensed by cooling to about 1100 K in steel-lined condensers, and then removed and cast into ingots:
The forward reaction is endothermic and the position of equilibrium is in favour of magnesium oxide. However, by removing the magnesium vapour as it is produced, the reactiongoes to completion. The silica combines with calcium oxide to form the molten slag, calcium silicate:
The process gives magnesium with up to 99.99% purity, slightly higher than from the electrolytic processes.
(b) The electrolytic process
Outside China, the electrolytic process is usually the preferred choice.
The process involves two stages:
i) production of pure magnesium chloride from sea water or brine
ii) electrolysis of fused magnesium chloride
(i) Production of pure magnesium chloride from sea water or brine
Where sea-water is the raw material, it is treated with dolomite which has been converted to mixed oxides by heating to a high temperature. Magnesium hydroxide precipitates, while calcium hydroxide remains in solution. Magnesium hydroxide is filtered off and on heating readily forms the pure the oxide.
Conversion to magnesium chloride is achieved by heating the oxide, mixed with carbon, in a stream of chlorine at a high temperature in an electric furnace (Figure 1).
Figure 1 Illustrating the production of magnesium chloride from magnesium oxide.
Several reactions occur:
Where magnesium chloride-rich brines are the source of magnesium, the solution is treated for removal of various impurities and the remaining magnesium chloride solution concentrated by evaporation in several stages.
The last stage of dehydration has to be carried out in the presence of hydrogen chloride gas to avoid hydrolysis of the magnesium chloride:
A new process is under development using magnesite. Small pieces of the ore are converted directly to molten magnesium chloride by heating with chlorine in an electric furnace in the presence of carbon monoxide.
(ii) The electrolysis of fused magnesium chloride
The resulting anhydrous magnesium chloride is fed continuously into electrolytic cells (Figure 2) which are hot enough to melt it.
On electrolysis, magnesium and chlorine are produced:
Figure 2 Illustrating the electrolysis of magnesium chloride.
The molten metal is removed and cast into ingots. The chlorine gas is recycled to the chlorination furnace.
Secondary production
Only about 3% of the total magnesium used annually is from recycling, an estimated 23 000 tonnes.
Date last amended: 2nd October 2016