Uses of propanone (acetone)
The plastics industry uses about half the propanone produced.
It can be converted into bisphenol A, used in the manufacture of polycarbonates and epoxy resins, used in paints and into methyl 2-methylpropenoate used to make poly(methyl 2-methylpropenoate),sold as Perspex.
It is converted into 4-methylpentan-2-one (methyl isobutyl ketone, MIBK) (see below) which is a very useful solvent for paints.

Figure 1 Uses of propanone.
Data for 2013 from: Grand View Research, 2016
The figures for the different proportions of propanone used to make other materials given in Figure 1 are for the World but they vary from country to country. For example, in the US the proportion of propanone used to make methyl 2-methylpropenoate rises to 50%, whereas that for solvents is only 20%. The proportion used to make bisphenol A is similar.
Annual production of propanone (acetone)
| World | 6.7 million tonnes1,2 |
| Asia | 2.7 million tonnes1 |
| US | 1.8 million tonnes1 |
| Europe | 1.7 million tonnes1 |
1. Merchant Research and Consulting, 2016
2. It is expected to be more than 8.4 million tonnes in 2017
Manufacture of propanone (acetone)
There are two ways of manufacturing propanone, one via cumene and one via propan-2-ol (isopropanol). Both of these intermediates are produced from propene. By far the most important route is the cumene process.
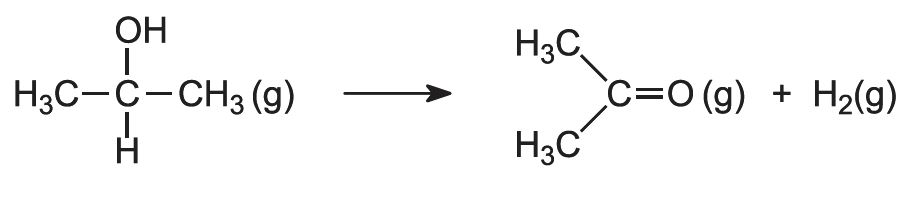
The second route is by dehydrogenation of propan-2-ol. The alcohol vapour is passed over zinc oxide at ca 600 K or copper at ca 750 K, which act as catalysts:
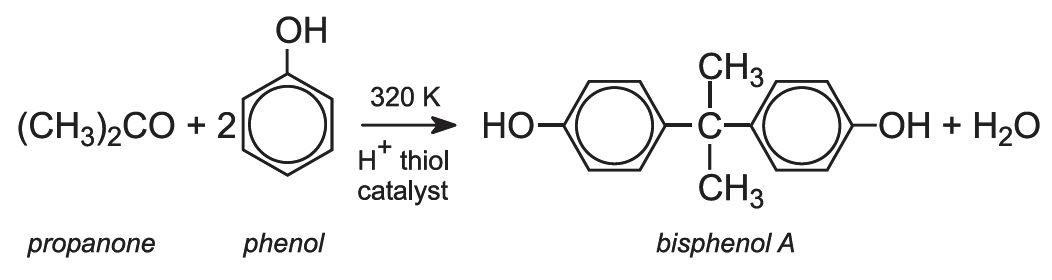
Bisphenol A
To make bisphenol A, the two products from cumene oxidation are reacted together:
In the past, hydrogen chloride was used as the acid catalyst, and some inevitably escaped into the atmosphere. Now a solid cation exchange resin is used which is more environmentally friendly.
Although the main use of bisphenol A is in the manufacture of polycarbonates, it is also used in the manufacture of fire retardants, including tetrabromobisphenol A.
4-Methylpentan-2-one (methyl isobutyl ketone, MIBK)
MIBK is produced from propanone:
Date last amended: 9th January 2017