.jpg) Ethene (ethylene) is the most important organic chemical, by tonnage, that is manufactured. It is the building block for a vast range of chemicals from plastics to antifreeze solutions and solvents.
Ethene (ethylene) is the most important organic chemical, by tonnage, that is manufactured. It is the building block for a vast range of chemicals from plastics to antifreeze solutions and solvents.
.jpg) Figure 1 On the site at Grangemouth in Scotland, ethene is produced by steam cracking of naphtha.
Figure 1 On the site at Grangemouth in Scotland, ethene is produced by steam cracking of naphtha.
By kind permission of INEOS Manufacturing Scotland.
Uses of ethene (ethylene)
The principal uses of ethene are to produce:
a) polymers
- poly(ethene)
- ethylbenzene and hence phenylethene and poly(phenylethene)
- chloroethene (vinyl chloride) and hence poly(chloroethene), i.e. poly(vinylchloride), PVC
- ethylbenzene and hence phenylethene and poly(phenylethene) polystyrene
b) other chemicals
- epoxyethane and hence the diols, such as ethane-1,2-diol
- ethanol
The manufacture of polymers is the main use of ethene. Poly(ethene) accounts for about 60% of the world demand for ethene (HDPE 28%, LLDPE 18%, LDPE 14%), while dichloro-1,2-ethane, the precursor for chloroethene and thence PVC, accounts for a further 11%. Ethylbenzene, the route to poly(phenylethene), uses another 5%1.
About 16% of ethene is used worldwide to make epoxyethane (ethylene oxide).
Annual production of ethene (ethylene)
| World | 134 million tonnes1 |
| Asia Pacific | 50 million tonnes1 |
| U.S. | 25 million tonnes2 |
| Europe | 20 million tonnes3 |
| Middle East | 20 million tonnes1 |
Data from:
1. Calculated from data, Nexant and ChemVision. Estimated data for 2014
2. 2015 Guide to the Business of Chemistry, American Chemistry Council 2016. Data for 2014
3. Petrochemicals Europe 2016. Data for 2015
Much of the ethene produced in the Middle East comes from Saudi Arabia, 17 million tonnes a year, with one refinery producing over 2 million tonnes a year, the 4th largest ethene production plant in the world.
Manufacture of ethene (ethylene)
Ethene is produced from the cracking of fractions obtained from distillation of natural gas and oil.
The processes are:
a)the steam cracking of ethane and propane (from natural gas and from crude oil)
b) the steam cracking of naphtha from crude oil
c) the catalytic cracking of gas oil from crude oil. The choice of feedstock depends on availability, price
(which can vary considerably), and what other products from cracking are needed.
The vast majority of ethene is produced by steam cracking. Some crackers are capable of producing 3 600 tonnes of ethene a day.
.jpg) Figure 2 Distribution of ethene by pipeline across Europe.
Figure 2 Distribution of ethene by pipeline across Europe.
Within Europe, the ethene that is not needed at the site at which it is produced is fed into pipelines which connect other chemical plants and refineries (Figure 2).
A new wave of crackers is being built in the United States to take advantage of a large supply of ethane and other hydrocarbon gases from fracking. The US produces about 25 million tonnes of ethene a year. In 2010, about 9 million tonnes came from ethane and another 4 million from propane. By 2015 the amount from ethane had increased to 10 million tonnes and by 2020 it is expected to rise to about 140 million tonnes from ethane and 50 from propane, a direct result of the growth in fracking. In 2016 the first shipments of ethene from fracking fields in the US were being processed in refinmeries in Europe.
|
New plants based on bioethanol produced from sugar cane are in operation and are also being constructed in Brazil. High yields of bio-based ethene are obtained by dehydrating ethanol vapour using a catalyst containing a mixture of magnesium oxide, alumina and silica at 600-750 K:
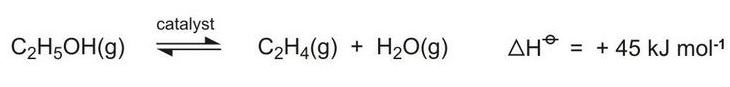
The ethene is used principally to produce bio-based poly(ethene).
Date last amended: 4th January 2017

