 The production of phenol from benzene is the first step in the manufacture of some very important polymers, including polyamides (nylons), polycarbonates and the phenolic resins.
The production of phenol from benzene is the first step in the manufacture of some very important polymers, including polyamides (nylons), polycarbonates and the phenolic resins.
Uses of phenol
The most important chemical made from phenol is bisphenol A, which is used to make the polycarbonates. Phenol is also catalytically reduced to cyclohexanol, which is used in the manufacture of polyamides 6 and 6,6.
Phenol is also used to make a range of thermosetting polymers (resins). It reacts with methanal in the presence of a catalyst to form phenol-methanal resins.
Figure 1 Uses of phenol.
Data for 2012, estimated from American Chemical Society, 2015
Among the other uses of phenol is the production of phenylamine (aniline) needed, for example, for the manufacture of dyes. Antiseptics such as 2,4-and 2,6-dichlorophenols are also made from phenol.
Annual production of phenol
| World | 8.9 million tonnes1,2 |
| Asia | 3.7 million tonnes1 |
| Europe | 2.7 million tonnes1 |
| US | 2.3 million tonnes1 |
1. For 2012, estimated from Merchant Research and Consulting, 2014
2. Forecast 10.0 million tonnes for 2015
Manufacture of phenol
The vast majority of phenol is made by the cumene process. The process has three stages:
a) production of cumene
b) conversion of cumene to cumene hydroperoxide
c) decomposition of cumene hydroperoxide
(a) Production of cumene
Cumene is the name often given to (1-methylethyl)benzene (isopropylbenzene). It is produced by the reaction of benzene and propene, using an acid catalyst; this is an example of a Friedel-Crafts reaction:
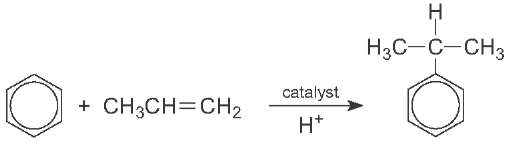
In one process, benzene and propene (3:1) are passed over an acid catalyst, usually a zeolite such as ZSM-5 at ca 600 K and under pressure (ca 10 atm) in a fixed bed reactor. The zeolite is more environmentally friendly than traditional acid catalysts such as aluminium chloride. The effluent is much cleaner and lower temperatures and pressures can be used.
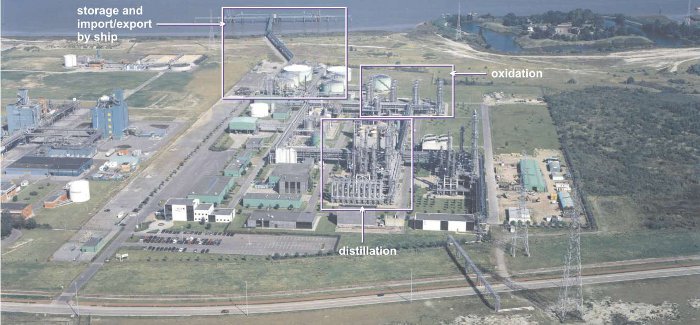
Figure 2 An aerial view of the plant producing phenol and propanone from cumene.
By kind permission of INEOS Phenol GmbH.
Alternatively, propene gas is liquefied under pressure (ca 30 atm) and mixed with benzene before being passed, still under pressure, through a reactor containing the solid zeolite at ca 435 K. This process is becoming more popular as it uses even lower temperatures and thus saves energy.
In some plants, solid phosphoric acid is used as the catalyst, in place of zeolites.
(b) Conversion of cumene to cumene hydroperoxide
Cumene is then oxidized with air to give the hydroperoxide (Figure 3). The reaction is autocatalyzed by cumene hydroperoxide. The overall reaction can be represented as:
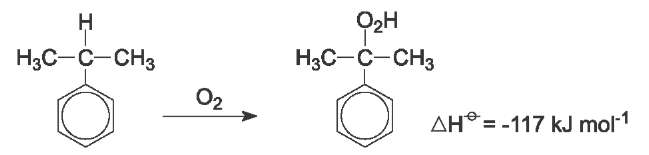
The reaction takes place at temperatures between 350-390 K and 1-7 atm pressure, the latter to retain the system in the liquid phase.

Figure 3 Oxidation units in which cumene hydroperoxide is produced.
By kind permission of INEOS Phenol GmbH.
At higher temperatures, the hydroperoxide is unstable and could decompose violently. The oxidizers are generally built some distance from the rest of the plant.
(c) Decomposition of cumene hydroperoxide
Finally, the hydroperoxide is mixed with sulfuric acid at 313-373 K to give, after neutralisation, phenol and propanone. This reaction when carried out with small amounts of sulfuric acid (500 ppm by mass) is termed homogeneous cleavage:
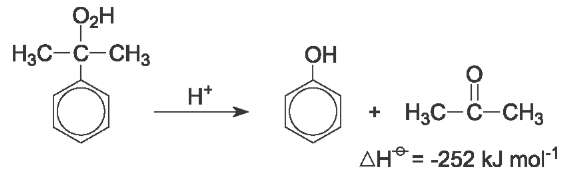
The products are separated by distillation, in up to six columns. Product yield is 85-87%, based on benzene.

Figure 4 Distillation units to separate phenol and propanone.
By kind permission of INEOS Phenol GmbH.
The Future
The cumene process has one problem. Six tonnes of propanone are produced for every ten tonnes of phenol. Thus, for economic efficiency, the manufacturer needs to sell the two compounds in that proportion. Often, much more phenol is needed than the propanone that is produced at the same time. There has been much research to find another route from benzene to phenol. One elegant method is by oxidation of benzene with nitrogen(I) oxide (nitrous oxide), N2O:
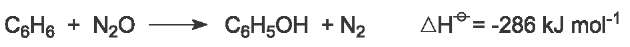
Nitrous oxide is a gaseous by-product in the production of polyamide 6,6 and cannot be allowed to escape into the atmosphere as it is a greenhouse gas. The reaction takes place in the gas phase at 570-720 K over a zeolite catalyst. 95% of the benzene is converted to phenol. Thus, here is a method by which a useful product can be produced from a by-product that is harmful.
Although the trials using the pilot plant were promising, no fully commercial plant has been built.
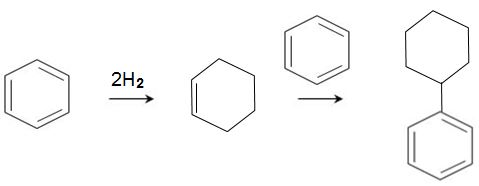
Another process, being developed by ExxonMobil, is very attractive as it also produces phenol from benzene without propanone.
In this process, benzene is hydrogenated to cyclohexene which reacts, over a zeolite impregnated with a transition metal (for example, palladium, ruthenium, nickel, cobalt), with another molecule of benzene to form cyclohexylbenzene:
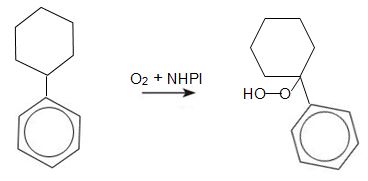
The cyclohexylbenzene is oxidized to a hydroperoxide. The overall equation is:
This is effected by NHPI (N-hydroxyphthalimide) which forms a radical which abstracts a benzylic hydrogen atom from cyclohexylbenzene, leading to the formation of the hydroperoxide.
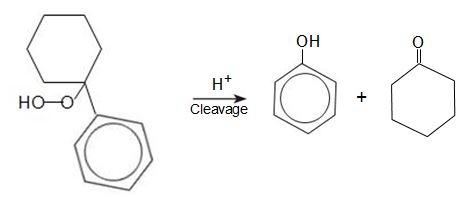
The hydroperoxide decomposes, in the presence of sulphuric acid, to form a molecule of phenol and a molecule of cyclohexanone which can be used in the manufacture of polyamides.
The process has similarities to the Cumene Process as a hydroperoxide is formed which is then decomposed by homogeneous cleavage to give two key products with very little unwanted side-products.
Date last amended: 11th January 2017