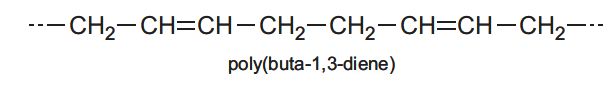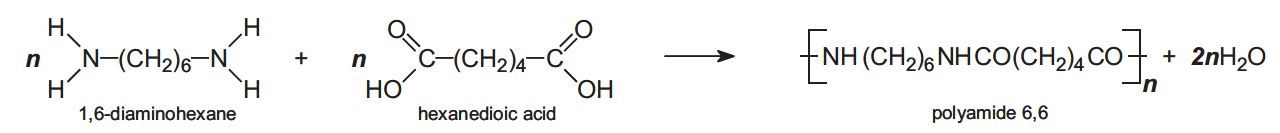When many molecules of a simple compound join together, the product is termed a polymer and the process polymerization. The simple compounds whose molecules join together to form the polymers are called monomers. The polymer is a chain of atoms, providing a backbone, to which atoms or groups of atoms are joined.
This unit provides an overview of the main types of polymers characterised by how they are made, how their structures govern their general properties and how these properties can be refined by their formulation using a range of additives. Finally the unit summarises the range of processing techniques that can be used to convert polymers into a vast range of different products.
Each of the other units in the Polymers section describes the manufacture, properties and uses of an individual polymer or group of polymers in more detail.
Polymers are large molecules, a type of macromolecule. Their chemical properties are similar to those of simple molecules. For example, if the polymer contains a carbon-carbon double bond, as in poly(but-1,3-diene), it will undergo additions reactions with, say hydrogen or bromine.
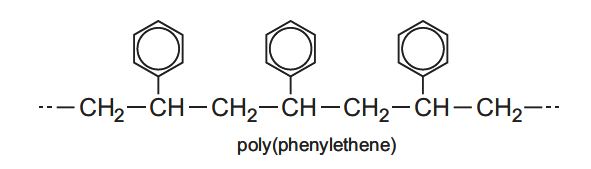
If it contains an aromatic ring, as in poly(phenylethene) (often known as polystyrene), it will undergo substitution reactions, say with nitric acid.
The major differences between smaller molecules and polymers lie not with their chemical properties but with their physical ones. Their larger sizes lead to much stronger intermolecular forces leading in turn to much higher melting points, and the characteristic properties of hardness and flexibility. These intermolecular forces are even stronger when the polymer chains pack together in a regular way as in HPDE (high density poly(ethene)) and have regions of crystallinity.
When heated, it melts and the crystallinity is lost. As it does not have a sharp melting point, the temperature at which this occurs is termed the melt transition temperature, Tm. Above this temperature, the polymer is amorphous.
Some polymers are hard and amorphous, having no regions of crystallinity, for example, poly(methyl 2-methylpropenoate). The temperature at which they become soft and pliable is termed the glass transition temperature, Tg.
Figure 1 These crystallites have order in which the zigzag polymer chains
are held together in a regular pattern by intermolecular forces.
There are many examples of polymers that occur naturally, for example, starch, cellulose and proteins. Over the last 70 years, synthetic polymers have been invented, often mimicking nature and they are now manufactured in millions of tonnes a year and are one of the most essential materials we use. Many are used as fibres. Others are moulded into required shapes and when they are used in this way, they are often termed plastics.
Characterising polymers
There are several ways in which polymers can be characterised:
a) how they are made, by addition or by condensation
b) whether they are homopolymers or heteropolymers (co-polymers)
c) whether they are themoplastics, thermosets, elastomers or fibres
d) by their steric structure
(a) Addition and condensation polymers
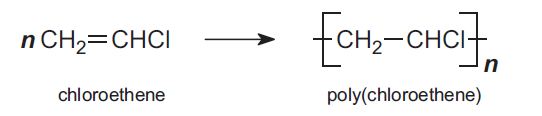
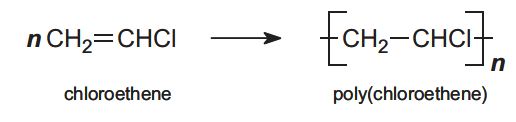
In addition polymerization, the polymer has the same empirical formula as the monomer but a higher molecular mass (Table 1). An example is the polymerization of chloroethene (vinyl chloride) to form poly(chloroethene), PVC:
| Monomer | Formula | Polymer | Trivial name | Structure |
|---|---|---|---|---|
| Ethene | 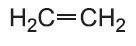 |
LDPE Low density poly(ethene) |
low density polythene |  |
| Chloroethene | 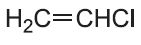 |
Poly(chloroethene) | polyvinyl chlorine, PVC | 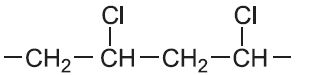 |
| Propene | 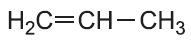 |
Poly(propene) | polypropylene | 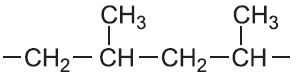 |
| Propenonitrile | 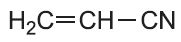 |
Poly(propenonitrile) | polyacrylonitrile | 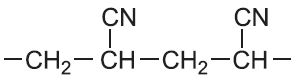 |
| Methyl 2-methylpropenoate | 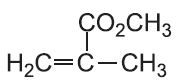 |
Poly(methyl 2-methylpropenoate) | polymethyl methacrylate | 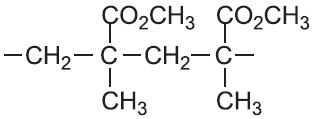 |
| Phenylethene | 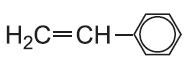 |
Poly(phenylethene) | polystyrene | 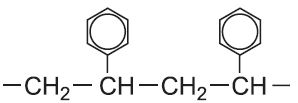 |
| Tetrafluoroethene | 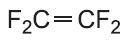 |
Poly(tetrafluoroethene) (PTFE) |
polytetrafluoroethylene PTFE |
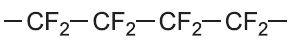 |
Table 1 Some addition polymers.
In condensation polymerization, polymerization of one or more monomers is accompanied by the elimination of small molecules (such as water or ammonia) (Table 2). For example, in producing polyamide 6,6, two monomers are used.
Another type of condensation polymer is said to be formed if the polymer chain contains (rather than appended to the chain) a functional group such as an ester, amide or urethane (Table 2).
| Polymer | Monomer | Formula |
|---|---|---|
| Polyesters | 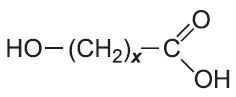 |
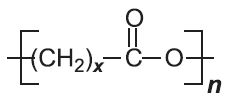 |
| Polyamides | 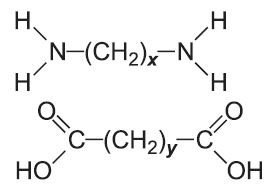 |
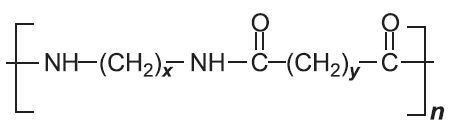 |
| Phenol-methanal plastics | 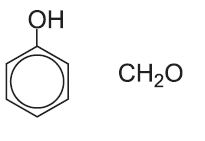 |
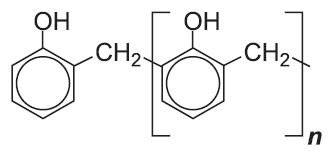 |
| Polyurethanes | 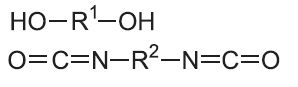 |
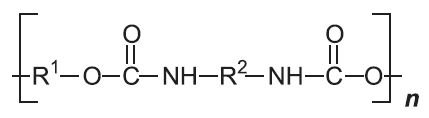 |
Table 2 Some condensation polymers.
(b) Homopolymers and heteropolymers (co-polymers)
Another way of characterising polymers is to divide them into homopolymers and heteropolymers. Many of the well known polymers such as poly(chloroethene) are produced from a single monomer and so are referred to as homopolymers (Table 1):
A heteropolymer, or as they are more commonly known, a co-polymer, is produced from two or more monomers.
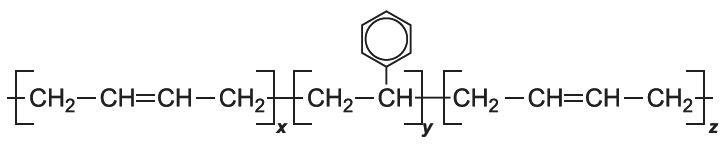
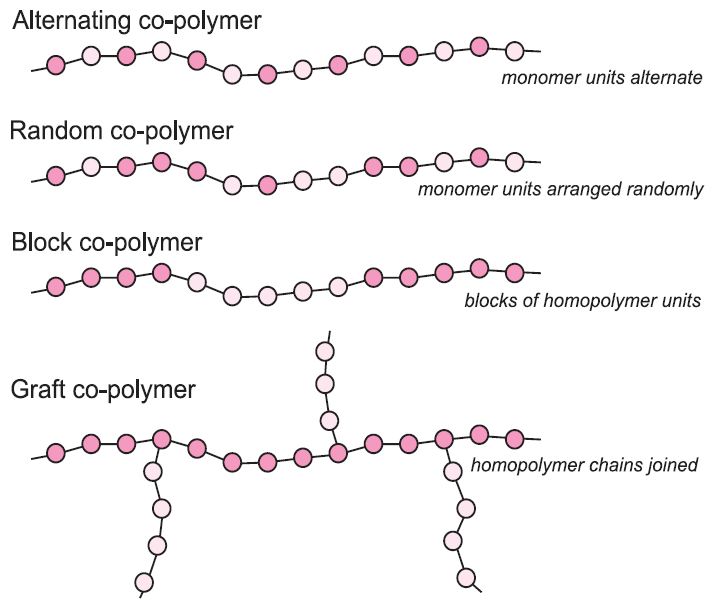
There are several types of co-polymer. One type is produced when two or more monomers are mixed and polymerized together. Depending on the reactivities of the monomers, they may form polymers with different arrangements of the monomer units (Figure 2).
SBS is an example of a block co-polymer. SBS is so named with S referring to Styrene (phenylethene) and B for Buta-1,3-diene. First, phenylethene is polymerized. Buta-1,3-diene is then added and adds on to both the reactive ends of the poly(phenylethene) molecules to form SBS:
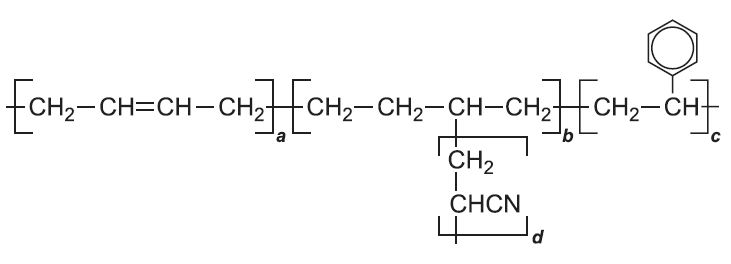
Another type of co-polymer is known as a graft co-polymer. An example is ABS. A is Acrylonitrile, the trivial name for propenonitrile.
The backbone of the polymer is formed from phenylethene (styrene) and buta-1,3-diene. Propenonitrile (acrylonitrile) is added to the system and forms a grafted side chain onto the backbone. The nitrile adds to the double bond on the butadiene unit:
Co-polymers are very useful as they have the properties of the constituent polymers and thus can be produced for specific purposes. For example, poly(phenylethene) (polystyrene) is brittle but when it is co-polymerized with buta-1,3-diene, the latter gives the polymer resilience and strength. Known as High Impact Polystyrene (HIPS), it can now receive impacts without damage.
Figure 2 Structures of different types of co-polymer.
|
Figure 3 Different types of co-polymer have different properties. On the right, the medical vials are made of a random co-polymer of ethene and propene which give a flexible and clear material. Below, the covering of the cable is a block co-polymer of the two alkenes, giving a very tough material with rubber-like properties. |
|
|
|
(c) Thermoplastics, thermosets, elastomers and fibres
Polymers can also be characterised into four classes:
- thermoplastics
- thermosets (thermosetting resins)
- elastomers
- fibres
Thermomplastics consist of individual molecules with no covalent bonding between them but held together by intermolecular bonding. The polymers become soft on warming and can be moulded. They can be repeatedly warmed, softened and remoulded. A list of examples is given in Table 1.
Thermosets, on the other hand, have many covalent bonds between the chains, leading to a three-dimensional structure, which can be regarded as a single molecule. They can be moulded by heat and pressure, but once moulded they cannot be remoulded. The most important examples include the plastics made from methanal (formaldehyde). Elastomers are amorphous solids which, as the name suggests, are elastic (Table 3). They have coiled chains which can be stretched out but spring back to their original shape when the stretching force is released.
Fibres are thin threads which are produced by extruding a molten polymer through a die in which there are small holes.
Fibres produced in this way include the polyamides (such as nylon), the polyesters (such as terylene) and poly(propene) (Table 4).
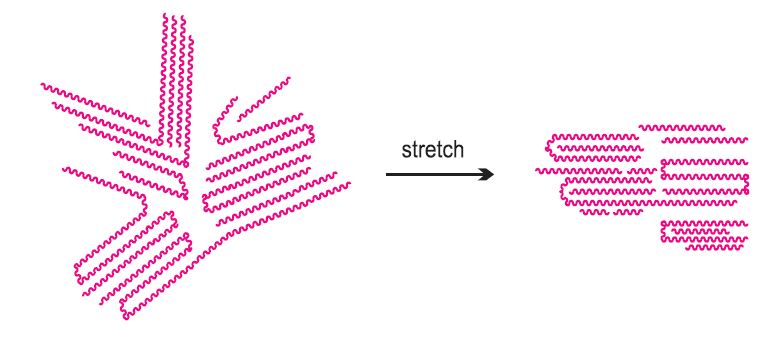
Figure 4 After stretching to make a fibre, the crystallite regions are
aligned along the axis of the fibre, and this adds strength to it.
Having been extruded and stretched, the polymer molecules become aligned in the direction of the fibre. Any tendency to return to a random orientation is prevented by the strong intermolecular forces between the molecules (Figure 4).
Fibres are twisted into threads and can then be woven into cloth or imbedded in a plastic to give it much greater strength (Table 4).
| Polymer | Formula |
|---|---|
| Poly(buta-1,3-diene) | 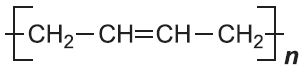 |
| Polyurethanes | 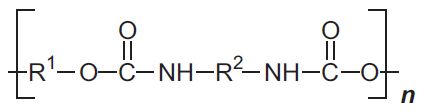 |
| ABS (Acrylonitrile butadiene styrene) |
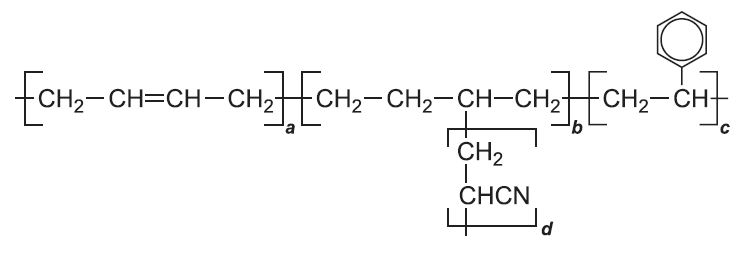 |
| Silicones | 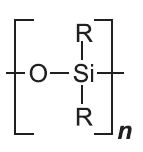 |
Table 3 Some elastomers.
| Polymer | Formula |
|---|---|
| Polyamide 6 | 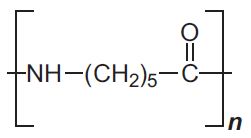 |
| Polyamide 6,6 | 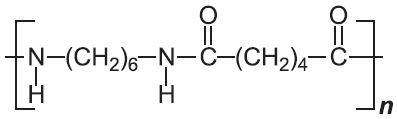 |
| The polyester (terylene) formed from the dimethyl ester of 1,4-benzenedicarboxylic acid and ethane-1,2-diol | 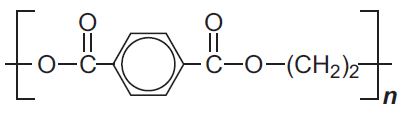 |
| Poly(propenonitrile) | 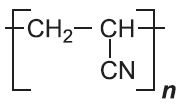 |
| Poly(ethene) | 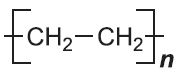 |
| Poly(propene) | 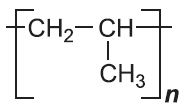 |
| Poly(chloroethene) | 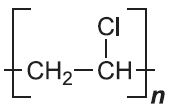 |
| Poly(tetrafluoroethene) | 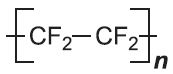 |
Table 4 Some polymers used to make fibres.
(d) Steric structure
Another way of classifying polymers is by examining their steric structure. Polymers with side chains can be divided into two classes, one (stereoregular) which has a recurring pattern in terms of stereochemistry and one (atactic) with no regular structure.
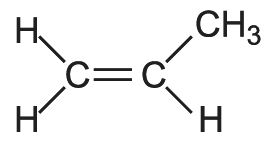
A simple example of a polymer with a side chain is poly(propene).
The propene molecule is asymmetrical,
and, when polymerized, can form three basic chain structures dependent on the position of the methyl groups: two are stereoregular (isotactic and syndiotactic) and the third does not have a regular structure and is termed atactic as shown diagrammatically below:
Figure 5 Molecular structures of poly(propene).
The 'one handed' structure of isotactic poly(propene) causes the molecules to form helices. This regular form permits the molecules to crystallize to a hard, relatively rigid material, which, in its pure form, melts at 440 K.
The syndiotactic polymer, because of its regular structure, is also crystalline.
Atactic chains are completely random in structure and consequently they do not crystallize. High molecular mass atactic poly(propene) is a rubber-like material.
Commercial poly(propene) is a predominantly isotactic polymer containing 1-5% by mass of atactic material.
Stereoregular polymers are produced when using many Ziegler-Natta (see below) or metallocene catalysts.
Manufacture of polymers
As discussed above, polymers can be characterised by the method of polymerization, addition and condensation.
Many addition polymers are produced by using an organometallic compound, known as a Ziegler-Natta catalyst. They were first developed by Karl Ziegler and Giulio Natta which led to the two chemists being awared the Nobel Prize in 1963 for this brilliant work.
Other addition polymers are produced by generating free radicals using a compound, known as an initiator, to catalyse the reaction.
Many condensation polymeriztion reactions, in which one or two monomers are the starting materials, also need catalysts. The catalysts are described in the unit devoted to each condensation polymer.
Catalysts for polymerization reactions
Ziegler-Natta catalysts
Ziegler-Natta catalysts are organometallic compounds, prepared from titanium compounds with an aluminium trialkyl which acts as a promoter:
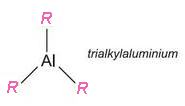
The alkyl groups used include ethyl, hexyl and octyl.
The role of the titanium catalyst can be represented as shown in Figure 6.
The alkene monomer attaches itself to an empty coordination site on the titanium atom and this alkene molecule then inserts itself into the carbon-titanium bond to extend the alkyl chain. This process then continues, thereby forming a linear polymer, poly(ethene).
The polymer is precipitated when the catalyst is destroyed on addition of water. Because it is linear, the polymer molecules are able to pack together closely, giving the polymer a higher melting point and density than poly(ethene) produced by radical initiation.

Figure 6 Illustrating the role of a Ziegler-Natta catalyst.
Not only do Ziegler-Natta catalysts allow for linear polymers to be produced but they can also give stereochemical control. Propene, for example can polymerize in three ways, as shown above in Figure 5, to produce either atactic, isotactic or syndiotactic poly(propene).
However, this catalyst only allows the propene to be inserted in one way and isotactic polypropene is produced.
Even greater control of the polymerization is obtained using a new class of catalysts, the metallocenes.
Radical polymerization
Many polymers, including all of the addition polymers in Table 1, are produced using radical initiators, which act as catalysts. For example the polymerization of chloroethene is started by warming it with a minute trace of a peroxide (R-O-O-R):
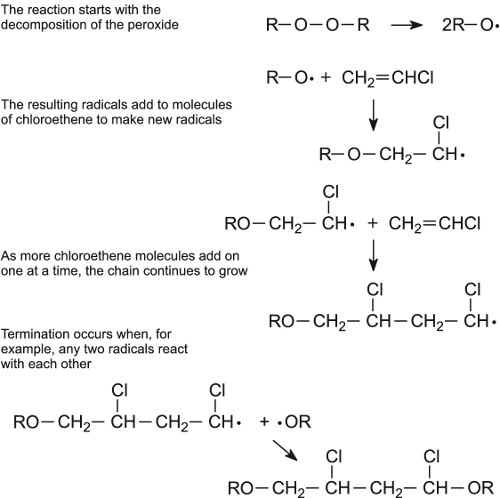
Figure 7 A mechanism for the free radical polymerization of chloroethene to poly(chloroethene).
In the case of ethene, by using the free radical process, the resulting polymer has a lower density and a lower softening point than the poly(ethene) produced using a Ziegler-Natta catalyst or a metallic oxide. The low density poly(ethene), LDPE, has side chains because the radicals react not only with molecules of ethene, by addition, but also with polymer molecules, by a process known as hydrogen abstraction. The polymer radical can also abstract a hydrogen atom from its own chain:
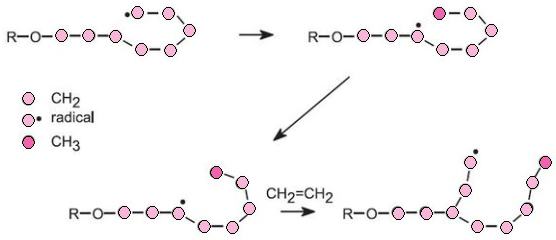
Both of these reactions lead to side chains so that the molecules of the polymer cannot pack together in a regular way. The polymer thus has a lower melting point and lower density.
Plastics formulation
The properties of many plastics can be modified by varying their formulation. For example, one of the most versatile plastics is poly(chloroethene) (PVC).
It can be made in either a flexible or a rigid form (and every combination between these extremes) by using various additives (Table 5).
| Figure 8 Specific properties can be produced by mixing polymers. For example this shirt is made from a mixture of poly(propenoate) (acrylic), aramid and polyamide fibres which gives protection against heat and yet remains comfortable to wear. By kind permission of DuPont. |
 |
| Additive | Examples | Function |
|---|---|---|
| Plasticiser | e.g. esters of benzene-1,2-dicarboxylic acid | Acts as a lubricant for polymer chains. Large amounts give a flexible product, low quantities produce a rigid one. |
| Stabiliser | e.g. lead carbonate (<1%), lead phosphate or, for non-toxic requirements, mixtures of metal octadecanoates and epoxidized oil | Prevents decomposition of polymer. Without a stabiliser, poly(chloroethene), for example, decomposes on heating to give a brittle product and hydrogen chloride. Some plastics become coloured (yellowing) when exposed to long periods of sunlight. |
| Extender | Chlorinated hydrocarbons | Extends the effect of the plasticiser, but generally cannot plasticise alone. They are cheaper than plasticisers, so help reduce costs. |
| Fillers | Chalk, glass fibre | Tailor the plastic for special requirements, or make it cheaper. |
| Miscellaneous | Flame retardants, UV stabilisers, antistatics, processing aids, pigments | Impart specially required properties to the plastic for the manufacturing process or for end-use. |
Table 5 Additives used to enhance the properties of plastics.
Processing plastics
Processing converts plastics into useful articles. Processing methods are given in Table 6.
| Process | Application |
|---|---|
| Compression moulding | Usually for thermosets - powder moulded under heat and pressure. |
| Injection moulding | Usually for thermoplastics - molten plastics injected into a mould under pressure. The mould surface detail can be accurately reproduced. Very widely used. |
| Rotational moulding | Usually for thermoplastics. The powder is heated in a closed mould which is rotated, fairly slowly, simultaneously about two axes. Surface detail is poor but this method can be used to make large hollow articles. |
| Reaction injection moulding | Usually for thermosets, polymerization takes place in the mould thereby producing the finished article directly from a resin. |
| Extrusion | Usually for thermoplastics - the molten plastics are fed by a screw through a die, which for sheet or film, for instance, is a slit. Various extensions to the process are possible - e.g. a tube may be inflated by air whilst still hot to produce a tubular film (for bags, etc.), or short lengths of hot extruded tube can be inflated in moulds to form bottles. |
| Calendering | Usually for thermoplastics - molten plastics squeezed between hot rollers to form foil and sheet. |
| Thermoforming | Heat-softened thermoplastic sheet is drawn into or over a mould. If a vacuum is used to 'suck' the sheet into a mould, the process is known as vacuum forming. This process is used for a variety of articles, ranging from chocolate box liners to acrylic baths. |
Table 6 Methods of processing plastics.
Polymers: their manufacture and uses
While this unit is concerned with the general principles underlying the structure, formulation and processing of polymers produced today, the manufacture and properties of the polymers vary considerably. The following are discussed in other units:
Poly(methyl 2-methylpropenoate)
Important developments in recent years include degradable plastics and methods of recycling polymers which include reusing the polymer and degradation followed by repolymerization.
Date last amended: 18th March 2013