Paint is used to decorate, protect and prolong the life of natural and synthetic materials, and acts as a barrier against environmental conditions.
Paints may be broadly classified into Decorative paints, applied on site to decorate and protect buildings and other objects, and Industrial coatings which are applied in factories to finish manufactured goods such as cars.
The constituents of paint
Paints contain:
- pigment(s) - prime pigments to impart colour and opacity
- binder (resin) - a polymer, often referred to as resin, forming a matrix to hold the pigment in place
- extender - larger pigment particles added to improve adhesion, strengthen the film and save binder
- solvent (sometimes called a thinner) - either an organic solvent or water is used to reduce the viscosity of the paint for better application. Water-borne paints are replacing some paints that use volatile organic compounds such as the hydrocarbons which are harmful to the atmosphere.
- additives - used to modify the properties of the liquid paint or dry film
The binder (resin) and solvent together are sometimes known as the vehicle. The binder may be dissolved as a solution or carried as a dispersion of microscopically small particles in a liquid.
Depending on the type of paint and intended use, additives may include:
- dispersants - to separate and stabilise pigment particles
- silicones - to improve weather resistance
- thixotropic agents - to give paints a jelly-like consistency that breaks down to a liquid when stirred or when a brush is dipped into it
- driers - to accelerate drying time
- anti-settling agents - to prevent pigment settling
- bactericides - to preserve water based paints in the can
- fungicides and algaecides - to protect exterior paint films against disfigurement from moulds, algae and lichen
Paints are formulated according to their proposed use - primer, undercoat, special finishes (matt, gloss, heat resistance, anti-corrosion, abrasion resistance). The pigment powder is broken down into individual particles which are coated by and dispersed in the binder (resin) - known as 'wetting out'. Solvent is then added to give the required consistency. Each batch of ingredients is thoroughly mixed in large, stirred containers with the required additives (Figure 1). Amounts ranging up to 40 000 dm3 of paint may be made in a single batch.
|
This unit discusses the most commonly used binders followed by the pigments.
Binders in paints
The three most important binders (resins) used in modern paints are:
- acrylic polymers (resins)
- alkyd polymers (resins)
- epoxy polymers (resins)
Acrylic polymers (resins)
The binder in many emulsion paints is based on homopolymers or co-polymers of ethenyl ethanoate (vinyl acetate) and a propenoate (acrylic) ester.
Ethenyl ethanoate is manufactured by passing a mixture of ethanoic acid vapour, ethene and oxygen over heated palladium(ll) and copper(ll) chlorides:
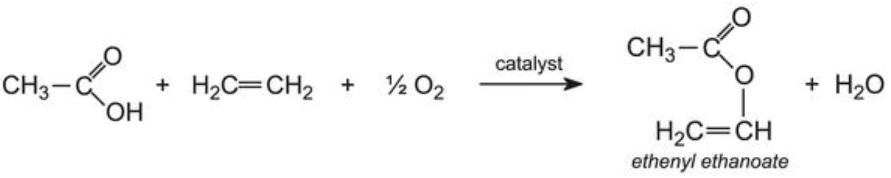
Ethenyl ethanoate and an acrylic ester (for example, methyl 2-methylpropenoate) are then co-polymerized to form a random array, in which these groups link into a linear chain:
.jpg)
Other acrylic esters used as co-monomers with ethenyl ethanoate are ethyl propenoate, butyl propenoates, or a co-polymer of butyl propenoate and methyl 2-methylpropenoate.
The polymers used in these paints are carried in water (water-borne emulsion paints) which as described above are much better for the environment than paints in which the binders are in organic solvents.
| Figure 2 Water-borne emulsion paints are used as decorative paints, particularly for the inside and outside of buildings (including masonry paints and exterior primers). By kind permission of AkzoNobel. |
 |
Emulsion paints are so-called as they are made by a process known as emulsion polymerization, in which the liquid monomers to be polymerized are first dispersed in water, as an emulsion. The polymers produced by this process typically have relative molecular masses of 500 000 - 1 000 000. As such they are useful only as dispersions since they would be extremely viscous if they were carried in solution and this would make them unusable.
.jpg)
Figure 3 Graph showing relationship between relative molecular
mass and viscosity for solution and dispersion polymers.
Acrylic resins may also be used in industrial paints, either as water-borne emulsion paints or as solvent-borne paints. Solvent-borne industrial paints can have a tough protective finish and are widely used in industry as topcoats, for example on car bodies. The paint frequently comes as two components which are mixed together just before use: the main paint portion typically consists of an acrylic resin produced by the polymerization of a propenoate ester formed from a polyhydric alcohol (diols and triols). The resulting polyester has numerous hydroxyl groups (-OH) pendant from the polymer backbone. The hydroxyl groups react with the other compound often consisting of a polymeric isocyanate such as a trimer of 1,6-diisocyanatohexane (hexamethylene diisocyanate):
.jpg)
Such a compound is known as a crosslinker for it produces, on reaction with the resin, a three-dimensional structure similar to the polyurethane formed from a polyol and an isocyanate.
When these two components are mixed together, a chemical reaction takes place between the hydroxyl groups on the polymer (acrylic resin) and the isocyanate groups on the cross linker:
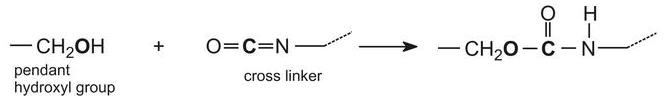
This reaction proceeds relatively slowly at room temperature, allowing enough time for the paint to be applied, after which the solvent thinner evaporates and the painted item is placed in an oven to accelerate the chemical reaction. This greatly increases the molecular mass of the polymer causing it to become a three-dimensional molecule and form a hard film, resistant to chemicals.
.jpg)
Alkyd polymers (resins)
Decorative gloss paints typically contain alkyd polymers (resins). A typical resin is that produced from a polyol such as propane-1,2,3-triol (glycerol) with a dibasic acid such as benzene-1,2-dicarboxylic (phthalic) anhydride and a drying oil (linseed or soybean oil). On being heated together, ester linkages are formed, and water is a by-product. The name alkyd is derived from alcohol and anhydride.
The first step in making the alkyd polymer is the reaction between the triol and the drying oil to produce a monoglyceride. For example:
.jpg)
The monoglyceride then reacts with the anhydride to form the alkyd polymer (resin):
.jpg)
The alkyd resins, which generally have relative molecular masses in the range of 10 000 - 50 000, are usually carried in organic solvents (solvent-borne paints). Turpentine extracted from trees was used in the past as the solvent, but this has been replaced by solvents from petrochemical feedstock, such as 'white spirit' which is a mixture of aliphatic and alicyclic hydrocarbons.
Once the alkyd resin is applied, the pendant oil drying groups react with oxygen in the air to form a cross-linked, hard thermoset coating, with a high molecular mass.
Epoxy polymers (resins)
Epoxy resins are often used as the binder in industrial coatings (primers). They give the paint excellent adhesion together with high resistance to chemicals (corrosion), and physical resistance necessary, for example, on ships and chemical storage tanks.
The epoxy resins are made from 1-chloro-2,3-epoxypropane (produced from 3-chloropropene) and substituted phenols, such as bisphenol A:
.jpg)
The value of n can be controlled to give a range of resins varying from viscous liquids to solids with high melting points. Epoxy resins can be carried in solvents such as aromatic hydrocarbons, alcohols, ketones and esters (solvent-borne paints) or as dispersions in water (water-borne paints) as true emulsions. They are not normally used in topcoats for outdoors because they are susceptible to UV degradation, but they make excellent interior coatings and exterior primers.
Epoxy resins are also used as adhesives (e.g. Araldite) and electrical insulators.
Pigments used in paints
Pigments give colour and opacity to paints. Amongst the organic pigments, particularly important are azo-, phthalocyanine and anthraquinone derivatives.
The most common inorganic pigment is white titanium dioxide (titanium(IV) oxide) which provides over 70% of total pigments used (Unit 51). It has a high refractive index and gives a 'gloss' to the paint. Another widely used inorganic pigment is finely divided calcium carbonate. This has a low refractive index and is used, together with titanium dioxide, to produce 'matt' paints. Other pigments include iron oxides (black, yellow and red), zinc oxide and carbon black.
Powdered metals such as zinc and some metal compounds, for example zinc phosphate, have corrosion inhibiting properties.
Paint drying
As the paint dries, a film is formed which adheres to the surface of the material to which it is being applied.
Emulsion paints dry by a physical process involving the evaporation of water followed by coalescence of the polymer droplets and their subsequent integration into a hard polymer matrix that acts as a binder for the pigment.
On applying gloss paints, the alkyd polymer cross-links by an oxidation reaction with oxygen in the air once the solvent has largely evaporated. This reaction is accelerated using salts of transition metals (for example, cobalt and manganese naphthenates). The transition metal ion (with variable oxidation state) catalyses cross linking of the polymer chains, producing a hard surface film to the paint.
Properties of an ideal paint
These vary greatly according to the particular end use. The requirements for an automotive topcoat, for example, will be very different to those for a decorative ceiling paint.
Some of the typical attributes required can include:
- ease of application
- good flow out of application marks (e.g. brush-marking)
- forming a continuous protective film
- high opacity
- quick drying
- corrosion resistance
- water resistance
- heat resistance
- colour stability (i.e. against visible and ultraviolet radiation)
- abrasion and scratch resistance
- durability
- flexibility
- easily cleaned
| Figure 4 These are weathering racks. Paints have been applied to panels and are exposed, at angles of 45° to the horizontal and south facing, to assess durability. Among the properties that are monitored are: colour change (fading), gloss change, dirt pick-up, cracking, flaking and contamination by fungi and algae. By kind permission of Q-Lab Europe Limited. |
|
Application methods
Numerous methods are used including: brush, roller, dipping, flowcoating, spraying, hot spraying, electrostatic spraying, airless spraying, electrodeposition, powder coating, vacuum impregnation and immersion.
Environmental issues
Lead compounds are no longer used in decorative paints and automotive paints. The quantity of lead compounds still being used in specialised industrial paints has been greatly reduced and eventually alternatives will be found. This also applies to chromates which, although they perform well and in the past have been extensively used on motor vehicles, are very toxic.
Because volatile hydrocarbons can lead to pollution in the troposphere, coatings with lower organic solvent content are required. The routes to achieve this include:
- water-based polymers (emulsion paints)
- higher solids content polymers (using less solvent)
- powder coatings
Water-based gloss paints are now available but the initial gloss of the finish is usually not as high as organic solvent based paints. The customer's choice is between a high performance product and a more environmentally friendly one. Intense research effort continues to improve these paints.
High solids paints (which are solvent-based) are now available but not without compromises in cost and performance. The relative molecular masses of the polymer resins are reduced to a maximum of ca 1000 compared to 5000 in conventional paints. This allows the proportion of the polymer to be increased from 20-30% to 40%, hence the term high solids. The main problem is the need to maintain a low viscosity. As the amount of solids increases so does the viscosity, reaching a point at which the paint cannot be applied properly. The lower proportion of solvent tends to slow down the drying and film hardening process, so changes are made to the structure of the polymer - increased branching tends to reduce viscosity for the same molecular mass. The application of the paint is more difficult. If applied by aerosol, the paint has to be under pressure. Sometimes the paint is applied hot. It is difficult to get as good a finish in appearance using a high solids paint.
|
Figure 5 Spraying a ship in dry dock. The lower part is often coated with paints containing a silicone (Unit 68) or a fluoropolymer (Unit 66) which prevent barnacles attaching to the ship and so reduces friction leading to reduced energy costs. A fouled ship can suffer a 40% increase in fuel consumption. |
 |
Powder coatings are used in particular for goods such as bicycles and white goods (refrigerators, washing machines). The powder is made up of a resin (often an epoxy resin), pigments, a catalyst to promote cross-linking when the powder is heated, and additives. The powder is sprayed on to the article using an electrostatic spray gun and is then heat cured to produce a hard coating. Recently acrylic powder coatings have been introduced as clear-coats on car bodies. Although an ideal solution for many applications, curing is achieved at high temperature in an oven and is therefore not universally applicable (e.g. painting of wood and plastics).
Date last amended: 18th March 2013

.jpg)
